PROJECTS
PROJECTS
Research Area A : Microglia

Research Area B : T cells

Research Area C : Human translation

Research Area Z : Core project (animal models)
Research Area A : Microglia
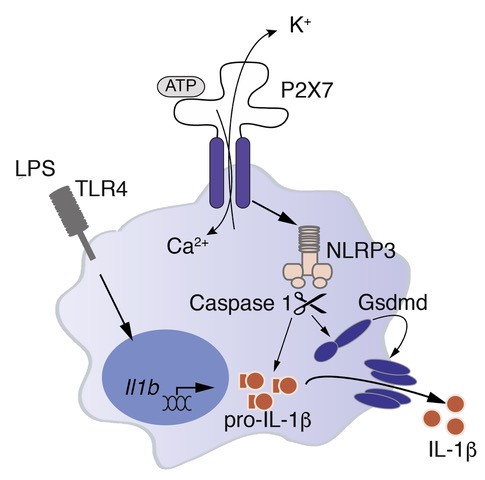
The main goal of this proposal is to analyze the role of GSDMD in acute stroke and its consequences for stroke recovery and evaluate GSDMD as a target for treament.
read more
We will use DMF and Disulfiram to modulate GSDMD in microglia and stroke-induced pyroptosis in the acute and during the recovery phase of stroke. To distinguish the pleiotropic effects of these drugs from effects on GSDMD in a cell specific manner, we will use inducible cell specific Caspase-1 knockout animals. Since no specific inhibitor for GSDMD exists, we plan to generate a nanobody against GSDMD which we hope to develop into a treatment option for stroke.

A2: Chronic inflammasome activation in post-stroke recovery
The objective of this project will be to assess the role of chronic lipid accumulation in inflammasome activation in the chronic phase after stroke, and analyze the impact of this mechanisms of functional recovery and neuronal plasticity after stroke.
read more
Our preliminary findings of an unexpected, pro-regenerative function after stroke suggest a potential involvement of Caspase-1 in currently unknown processes of tissue remodeling, neuronal plasticity or glia-neuronal interaction. These finding warrant a further in-depth analysis of the relevant cellular source of Caspase-1 in the chronic post-stroke phase, the mechanism of chronic inflammasome activation and its contribution to neuronal regeneration after stroke.
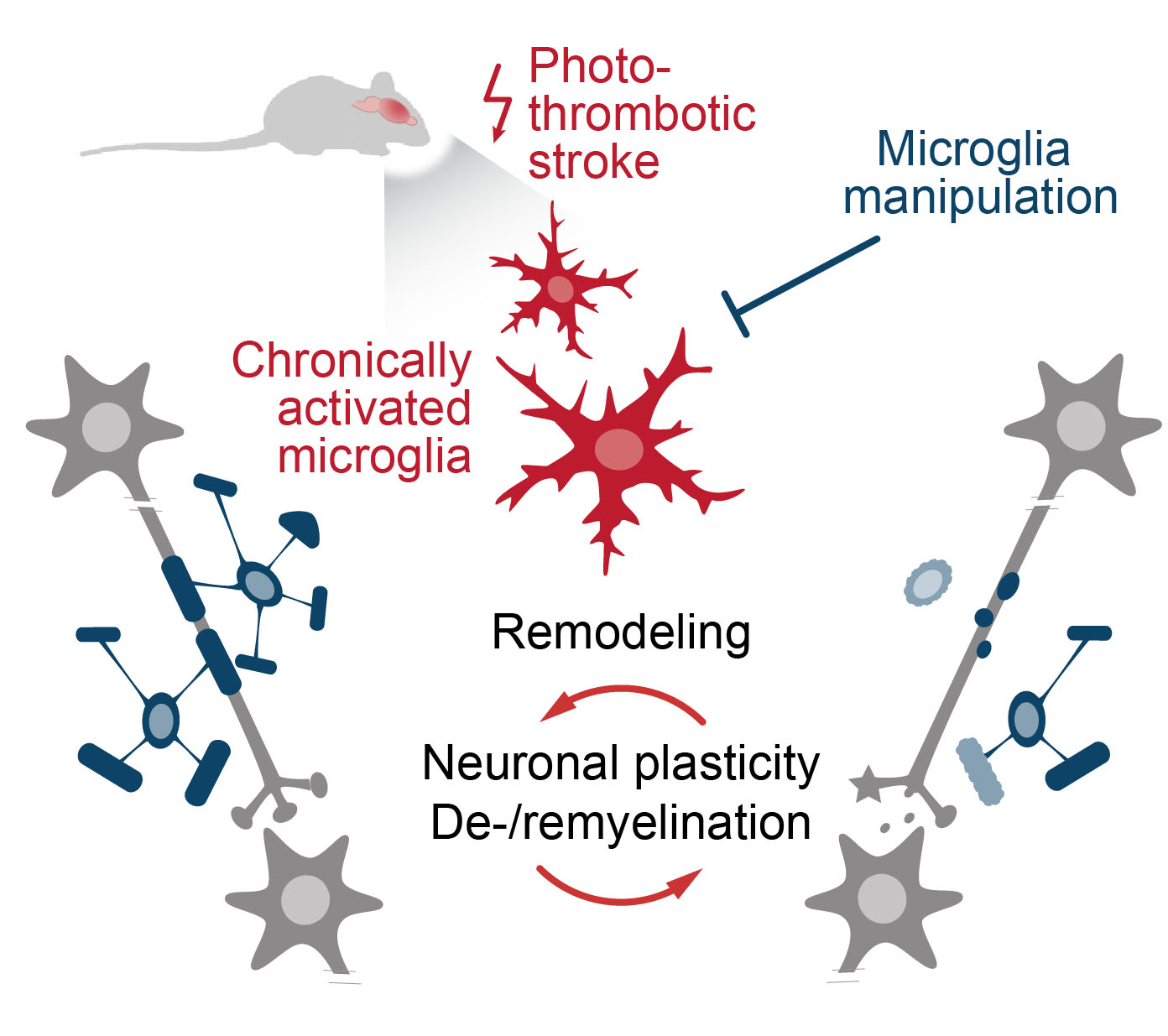
A3: Mechanisms of microglia-induced circuit remodeling in post-stroke recovery
The objective of this project will be to analyze the mechanisms by which microglia promote recovery after stroke, with an expanded focus on the microglial control of glial mechanisms of recovery, such as remyelination, during the chronic phase after stroke.
read more
We have recently found that microglia-mediated clearance of myelin sheaths is a key player of cortical remyelination; this study, however, only addressed the role of homeostatic microglia and did not involve ischemia. Thus, the impact of chronically activated microglia on remyelination post-stroke has yet to be studied in depth, and such an investigation will complement our ongoing work on microglial impact on multiple aspects of CNS remodeling.
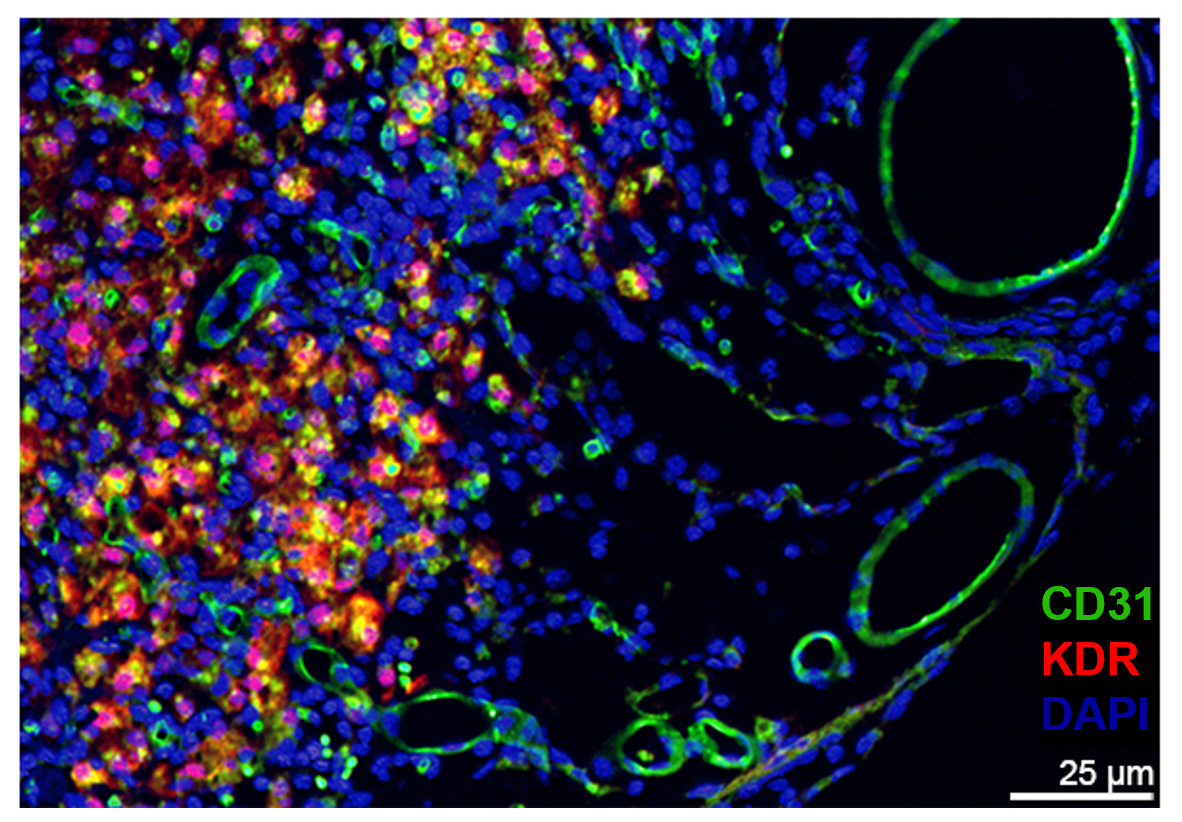
A4: Microglial responses controlling post-stroke angiogenesis and microvascular integrity
This project examines the hypothesis that microglia protects the cerebral microvasculature by promoting microvascular integrity and inhibiting leukocyte infiltration in the brain via mechanisms, which are at least partly ApoE-dependent.
read more
We will utilize our newly established platform of combined in vivo MRI/MPI and ex vivo LSFM from the first funding period for examining post-stroke microvascular network remodeling under microglial control.
Associated Project
Microvascular thrombosis as a novel therapeutic target for ischemic stroke
Our main objective is to study the inflammatory processes that occur in the ischemic-reperfusion area and are associated with spontaneous microthrombosis. This will allow us to investigate the role of microglia in the formation of microclots and the modulation of the blood-brain barrier (BBB) integrity. We want to understand how these processes may affect the outcome of a stroke.
read more
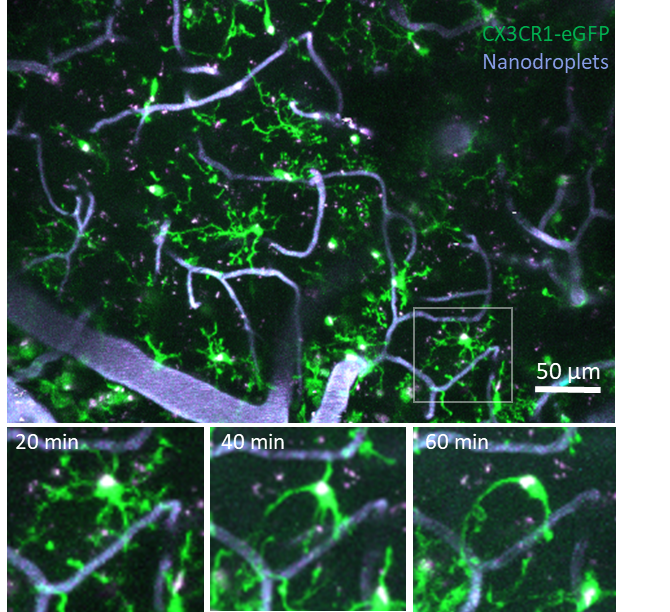
Using multiphoton microscopy, we will directly investigate microvascular thrombi, to unravel the time course of the no-reflow phenomenon and to explore the reaction and role of microglia
Research Area B: T cells
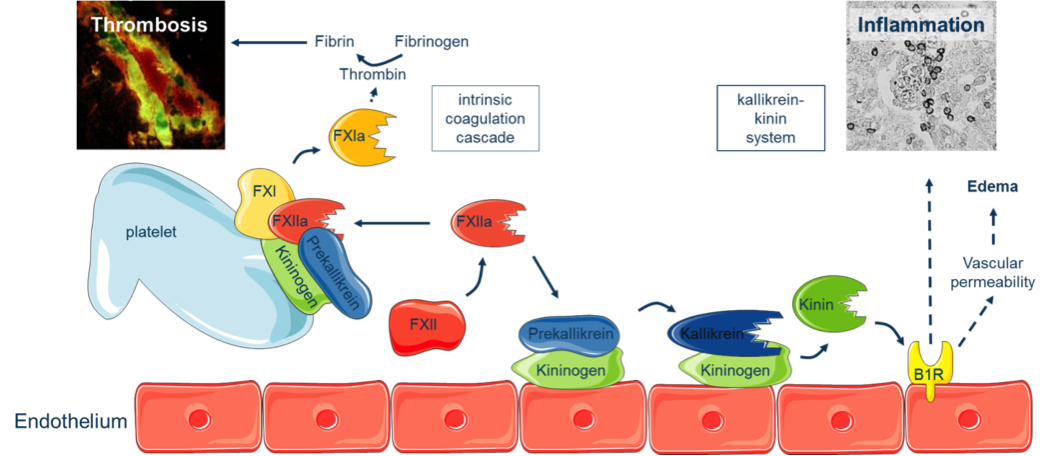
B1: Plasma-kallikrein mediated modulation of T cells and microglia responses during long-term stroke recovery
Therefore, the main objective of this project focuses on the characterization of how PK inhibition modulates T cell and microglia function upon brain ischemia together with the identification of mechanistically related targets using systems medicine in silico prediction.
read more
Our preliminary data suggest that: (i) PK inhibition enhances long-term stroke recovery by reducing the numbers of brain infiltrating T cells and microglia, (ii) PK might modulate T cell function in stroke via PARs, and (iii) a KKS-related disease module could identify de novo therapeutic targets beyond PK potentially targetable through pharmacological modulation.
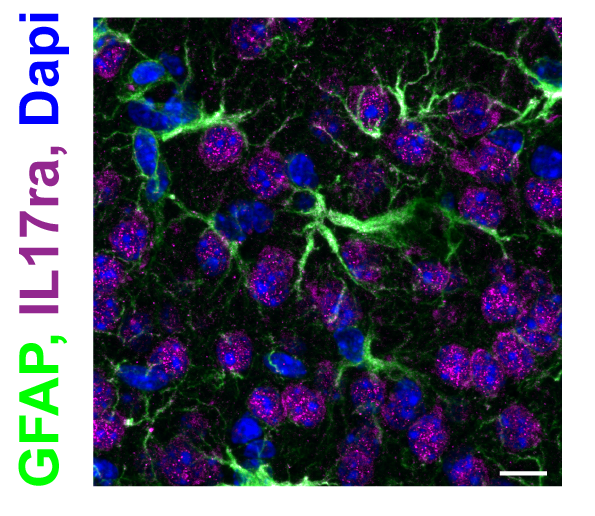
B3: Decoding IL-17A-mediated influences on stroke recovery
The overall objective of this project is to understand how the highly conserved cytokine IL-17A dictates stroke outcome. In extension to our neuron-focused approach in the first funding period, we will broaden our scope to reactive astrocytes.
read more
Specifically, we aim to decipher how IL-17A modulates neuronal recovery in the ischemic brain by directly engaging neuronal and astrocytic IL-17 receptors. Moreover, we will explore the contribution of astrocytes to stroke outcome by secreting IL-17A.
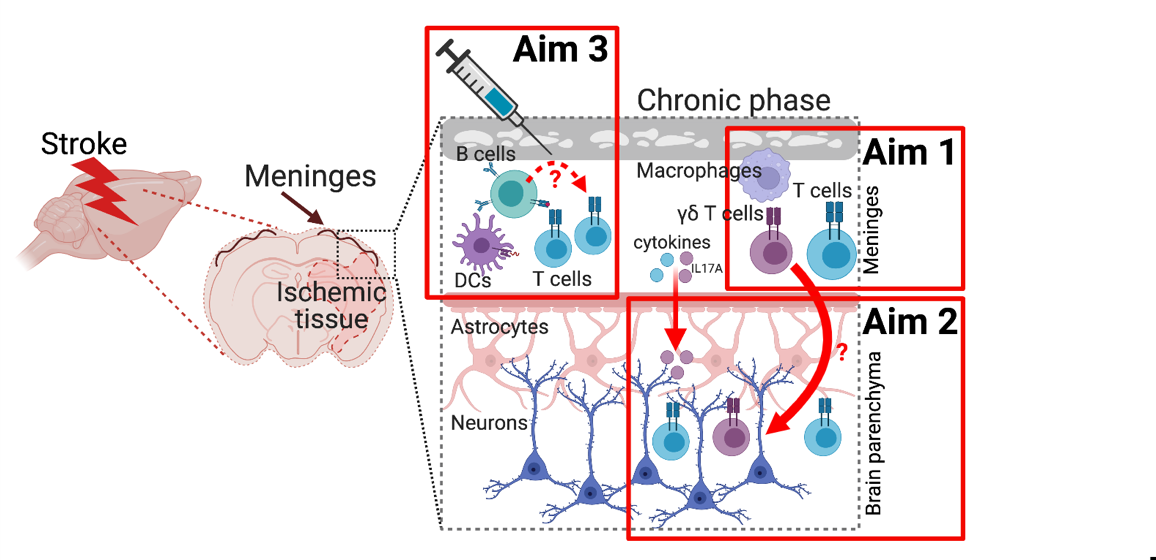
B4: Meningeal immune cell priming for stroke recovery
Here, we propose that the meningeal compartment represents an immune cell hub to influence chronic neuroinflammation and recovery mechanisms after stroke. Our preliminary data suggest that: (i) There is an accumulation of distinct leukocytes in the meninges in chronic response to brain ischemia, (ii) there are compartment-specific changes of meningeal immune cells after stroke and (iii) T lymphocytes are enriched in the extravascular dural space
read more
Hence, the key objective of this proposal is to investigate whether meningeal immune cells can influence the cerebral neuroinflammatory micromilieu and modulate sensorimotor recovery in the chronic response to stroke.
Research Area C: Human translation
C1: Linking functional immune profiles and ischemic lesion characteristics in human stroke
The goal of this project is to elucidate the dynamics of stroke-related changes in peripheral blood immune signatures (deep immune phenotyping approach) and functional properties of immune cells with a particular focus on myeloid cell populations, and to evaluate these changes in detail in relation to stroke subtype, characteristics of ischemic stroke lesions and disease outcome.
read more
We hypothesize that (1.) subtle alterations occur in the peripheral immune system after stroke, (2.) these alterations are related to stroke lesion characteristics, and (3.) they are associated with clinical recovery of the patients.
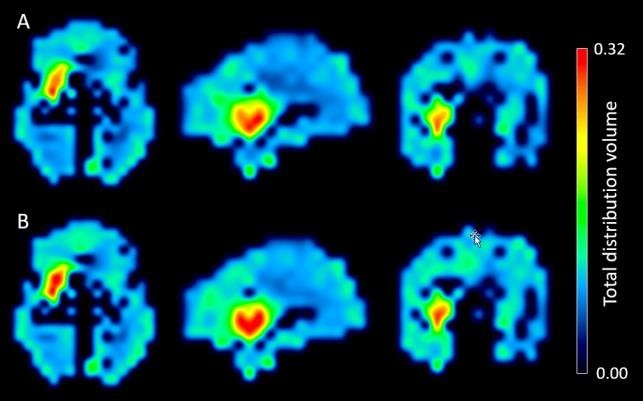
C2: Microglia-PET as a surrogate marker for post-stroke neuroinflammation
The goal of the current proposal is to address the first two hypotheses, to prepare for a future immune intervention trial by developing criteria for the selection of suitable patients using microglia-PET, and to explore feasibility of astrocyte-PET in experimental stroke. A novel radiosorting approach will be used to determine PET tracer sources at cellular resolution. This backtranslation will facilitate understanding of PET tracer signals. We expect these investigations to aid the interpretation of findings obtained in research areas A and C and facilitate transfer of novel therapeutic concepts into humans.
read more
Our overarching hypotheses are i) that a subpopulation of patients with acute stroke develop prominent microglial activation (MGA), ii) that patients with extensive MGA are more likely to have poor outcome and iii) that these patients are more likely to benefit from immune interventions.
Research Area Z: Core project (animal models)
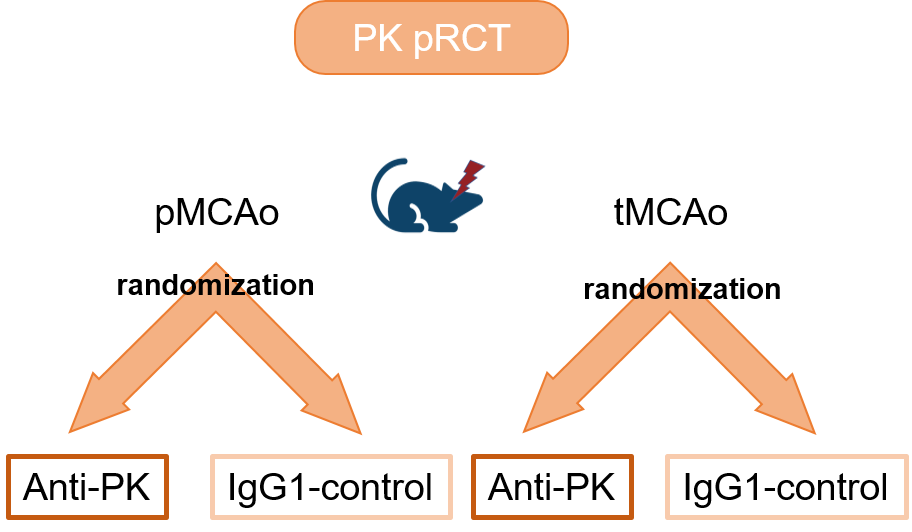
Z2: Preclinical RCTs and animal models support
The goal of this project is to maintain the high degree of harmonization of stroke models and readout parameters across ImmunoStroke laboratories by actively training all experimental surgeons and to perform two independent fully powered pRCTs to test the efficacy of a therapeutic candidates on post-stroke recovery.
read more
The work program includes an independent pRCTs as well as continuous full support to ImmunoStroke laboratories from the consortia groups in terms of personal training, sample providing and prepublication crosscheck.
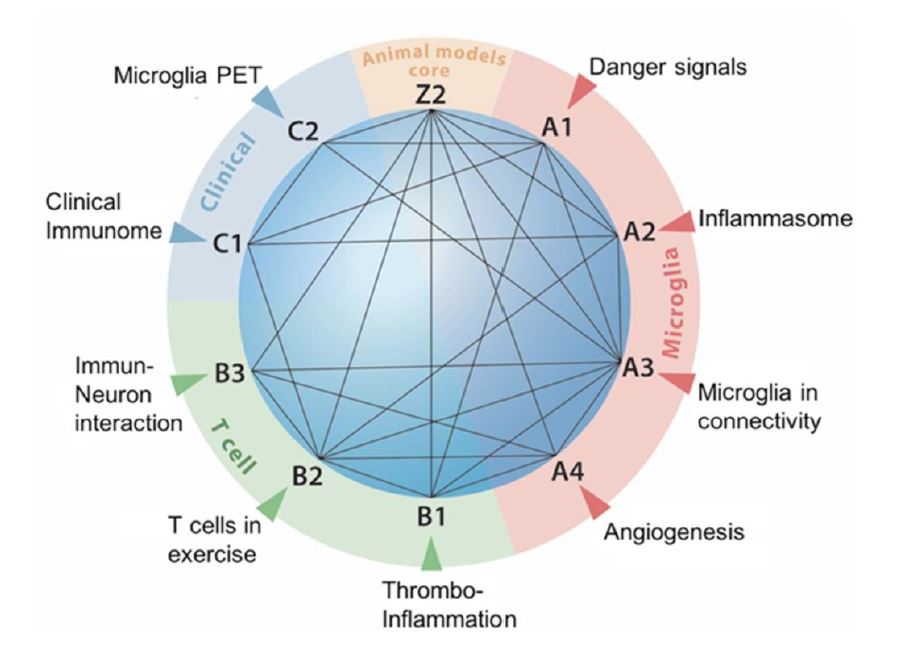
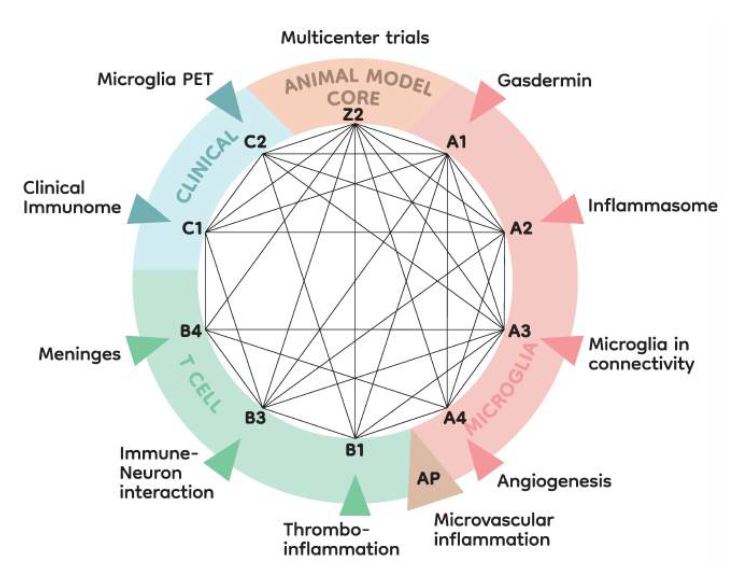
Research areas and interaction of projects
Our research group will investigate mechanisms of
regeneration and neuronal plasticity in the chronic phase after stroke with a focus on inflammatory mechanisms
driven by microglia and T cells. Accordingly, research Area A will investigate microglia-brain interactions while T
cells will be in the focus of research Area B. These two preclinical research areas will be complimented by two
clinical observatory studies in research Area C and a core project (Z2) for standardization of animal models and
outcome parameters.
In the second funding period, the overall structure of our consortium was maintained. We kept the three research areas: microglia (research Area A), T cells (research Area B) and clinical studies (research Area C). In addition, the Z02 project will conduct two pRCTs while maintaining the animal models platform.
Research areas and interaction of projects. In continuation of the structure of ImmunoStroke established in the first funding period, Area A will investigate microglia-brain interactions while T cells will be the focus of research Area B. These two preclinical research areas will be complemented by two clinical observatory studies in research Area C and a core project (Z2) for pRCTs and animal models platform. After positive evaluation, a topically related project (AP) will be associated.